Indole is a privileged nitrogen heterocycle and is found in structural scaffolds of many biologically active natural products, pharmacologically important agents and organic materials. Among the various indoles, bis(indolyl)methanes (BIMs) are valuable and ubiquitous in numerous natural resources and synthetic compounds. BIMs exhibit a variety of biological properties such as anti-bacterial, anti-oxidative, cytotoxic, anti-tumor, and anticancer activities. In the present work, bis(indolyl)methanes (BIMs) were readily prepared via the electrophilic substitution reaction of indoles with benzaldehydes mediated by hydrochloric acid supported on silica gel. The advantages of the present method included short reaction times, high yields, ease of experimental method and product isolation, benign reaction conditions and low cost.
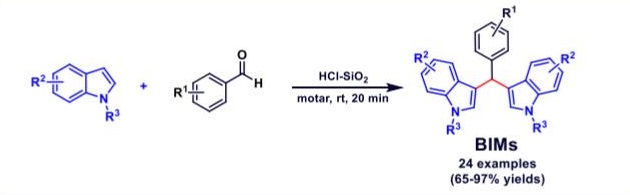
bis(indolyl)methanes catalyzed by HCl/silica gel under solvent-free conditions” Arkivoc 2021, viii, 190-203.
