Thanchanok Ratvijitvech* Department of Chemistry, Faculty of Science, Mahidol University
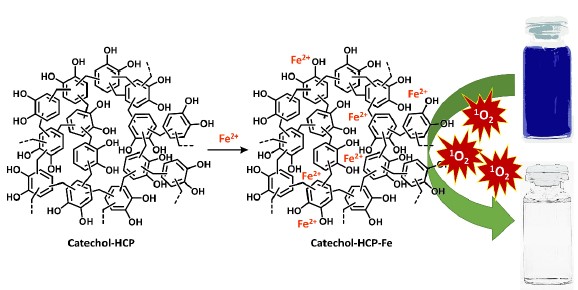
Clean water is one of the sustainable development goals. Organic dye is one of the water pollutants affecting water quality. Hence, the conversion of dyes to safer species is crucial for water treatment. The Fenton reaction using Fe as a catalyst is a promising process. However, homogeneous catalysts are normally sensitive, difficult to separate, and burdensome to reuse. Therefore, a catechol-based hypercrosslinked polymer (catechol-HCP) was developed as an inexpensive solid support for Fe (catechol-HCP-Fe) and applied as a heterogenous Fenton catalyst. The good interaction of the catechol moiety with Fe, as well as the porous structure, simple preparation, low cost, and high stability of catechol-HCP, make it beneficial for Fe-loading in the polymer and Fenton reaction utilisation. The catechol-HCP-Fe demonstrated good catalytic activity for methylene blue (MB) degradation even in a neutral pH. Complete decolouration of 100 ppm MB could be observed within 25 minutes. The rate of reaction was influenced by hydrogen peroxide concentration, polymer dose, MB concentration, pH, and temperature. The catechol-HCP-Fe could also be reused. By using abundant and low-cost chemicals and simple preparation methods, together with its good catalytic property with reusability, catechol-HCP is considered as an inexpensive material for metal ion nanoconfinement and heterogenous catalyst in Fenton reaction. Therefore, catechol-HCP is a promising candidate as an efficient and inexpensive novel material for wastewater treatment.
This research is supported by Mahidol University (Basic Research Fund: fiscal year 2021), grant number BRF1-A25/2564; and the CIF and CNI Grant, Faculty of Science, Mahidol University, grant number CH-TR:4/64#002.
Reference: “Fe-Immobilised Catechol-Based Hypercrosslinked Polymer as Heterogeneous Fenton Catalyst for Degradation of Methylene Blue in Water” Ratvijitvech, T. Polymers, 2022, 14 (13), 2749. https://doi.org/10.3390/polym14132749
