Institute: Department of Chemistry, Faculty of Science, Mahidol University.
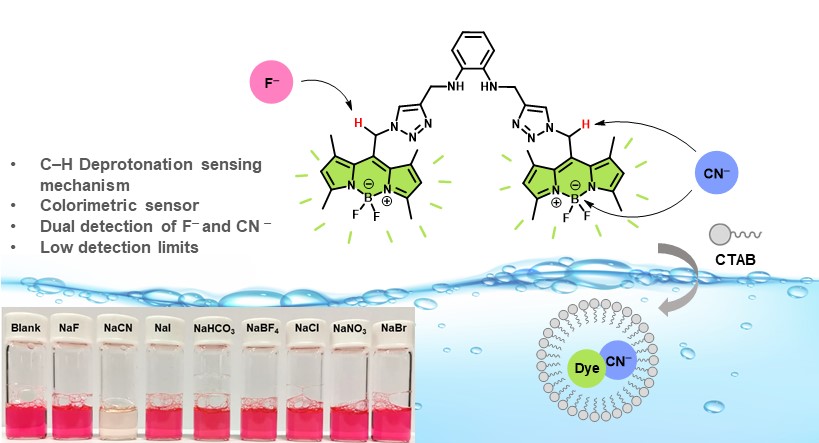
Research Background and its significance: A fluorescent probe for recognition of multiple target ions has the advantages of being cost-effective and highly efficient. In this work, bis-BODIPY-based colorimetric and fluorescent sensor (BODIPY–NN) prepared by bridging two units of BODIPY fluorophore/triazole binding group with a phenylenediamine unit. Its spectroscopic properties were investigated. BODIPY–NN presented “turn-off” fluorescence behaviors toward the F– and CN– ions over other interfering anions.
Scope of Research: This work focused on the development of a bis-BODIPY-based colorimetric and fluorescent sensor (BODIPY–NN) for F– and CN– recognition prepared by click chemistry and investigation of their selectivity and sensitivity toward anions ions by UV-VIS absorption and fluorescence spectroscopy methods.
Objective: To develop a new bifunctional fluorescence sensor for detection and differentiation of fluoride and cyanide ions.
Research grant: This research has received funding support from the NSRF via the Program Management Unit for Human Resources & Institutional Development, Research and Innovation (grant number B05F640128), the Faculty of Science, Mahidol University, the Center of Excellence for Innovation in Chemistry (PERCH-CIC), Ministry of Higher Education, Science, Research and Innovation, and Science Achievement Scholarship of Thailand (SAST).
Collaboration: Department of Chemistry and Center of Excellence for Innovation in Chemistry, Faculty of Science, Mahidol University.
Stakeholders: Scientific Community
Output/outcome: Publication/ a new fluorescent probe for fluoride and cyanide detection.
Web link: https://www.sciencedirect.com/science/article/pii/S0143720822005654
Graphical abstract:
Reference:
W. Saiyasombat, U. Eiamprasert, T. Chantarojsiri, K. Chainok, S. Kiatisevi, Bis-BODIPY-based fluoride and cyanide sensor mediated by unconventional deprotonation of C−H proton, Dyes Pigm. 206 (2022) 110643. https://doi.org/10.1016/j.dyepig.2022.110643.
